The biopharmaceutical airflow pulverizer is a specialized equipment for sterile ultrafine pulverization of biopharmaceutical raw materials such as antibodies, proteins, peptides, vaccines, etc. It uses non-contact low-temperature pulverization as the core and can process materials into submicron to micrometer sized powders, meeting the purity, activity, and particle size requirements of biopharmaceutical preparations (injections, freeze-drying agents, microspheres, etc.), and complying with pharmaceutical compliance standards such as GMP and FDA.
Biopharmaceutical airflow pulverizer
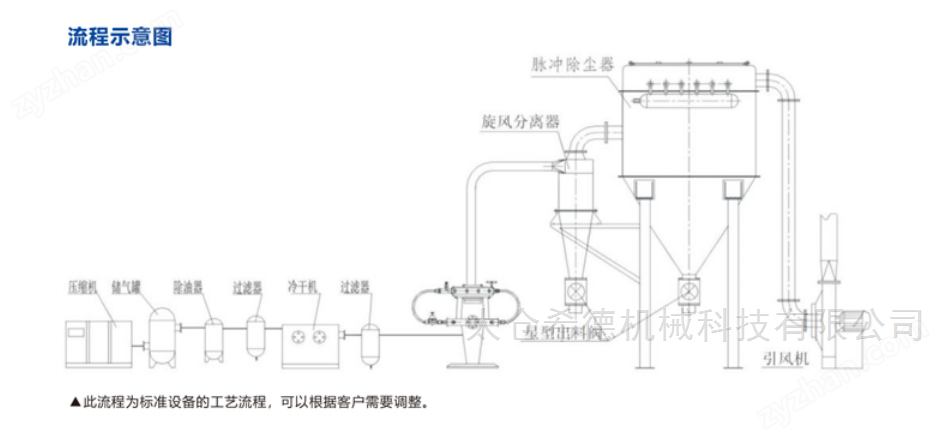
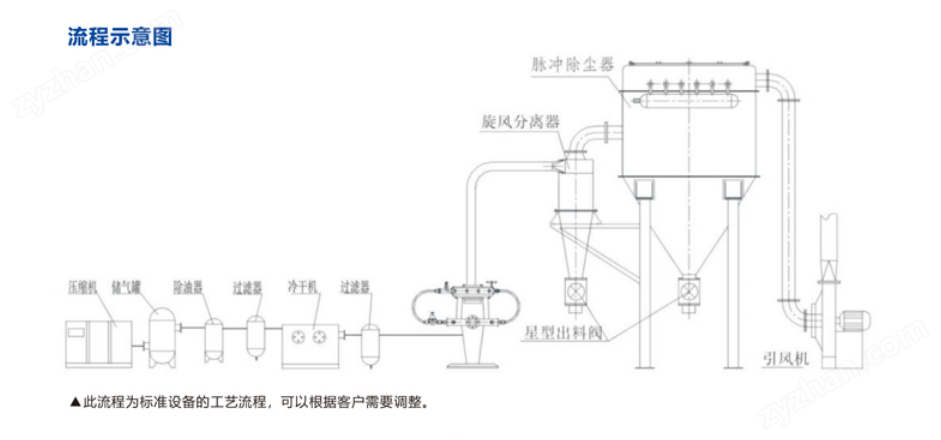
Working Principle:
It is achieved by compressing air through freeze-drying and filtering, allowing clean air to enter the crushing chamber at supersonic speed through the crushing nozzle, making the material in a fluidized state. The accelerated material converges at the intersection of several nozzle jet streams, producing intense collisions, friction, and shear to achieve ultrafine particle crushing. The material enters the grading zone with the rising airflow, and due to the high-speed rotation of the grading wheel, the crushed particles are simultaneously subjected to the centrifugal force of the grading wheel and the centripetal force of the fan, achieving the screening effect. Particles that meet the fineness requirements enter the next collection step through the gap of the grading wheel, while particles that do not meet the requirements return to the crushing chamber for further crushing.
Protein/antibody drugs: ultrafine grinding of monoclonal antibodies and fusion protein raw materials, used for injection freeze-dried powders and microsphere formulations to enhance dispersibility and stability.
Peptides/Vaccines: Aseptic crushing of peptide raw materials and vaccine adjuvants to ensure activity and purity, suitable for the production of GAO end injections.
Biological fermentation products: low-temperature crushing of thermosensitive materials such as enzyme preparations and probiotics, retaining biological activity, used for oral or topical preparations.
Gene therapy vector: ultrafine grinding of trace materials such as plasmids and viral vectors to avoid activity damage, suitable for gene therapy drug development and small-scale production.
Pre treatment: The moisture content of the material should be ≤ 3% to avoid sticking to the cavity; Priority should be given to using nitrogen protection mode for biologically active materials to prevent oxidation.
Parameter optimization: The intake pressure is 0.6-1.2 MPa, and the speed of the grading wheel is adjusted according to the target particle size; Live bacteria/protein materials can reduce the feeding rate and prolong the crushing time.
Cleaning validation: CIP cleaning after each batch, regular SIP sterilization (121 ℃, 30 min), retention of cleaning/sterilization records, in compliance with GMP data traceability requirements.
Equipment maintenance: Regularly check nozzle wear, grading wheel dynamic balance, replace seals to ensure crushing efficiency and sterility.

Research and development/small-scale testing: Select a laboratory small-scale machine (processing capacity of 0.2-5 kg/h), equipped with an inert gas system and online particle size monitoring, suitable for trace precious samples.
Pilot/mass production: Select models (10-50 kg/h) or large models (≥ 100 kg/h), support CIP/SIP and PLC automation control, and meet GMP batch production requirements.
Equipment selection table for biopharmaceutical airflow pulverizer:
parameter\\Model number |
SGN-300 |
SGN-400 |
SGN-600 |
SGN-800 |
SGN-1000 |
processing capacity(kg/h) |
50-260 |
260-600 |
500-1000 |
1000-3000 |
1500-3000 |
Air energy consumption(m³/min) |
1.25,0.7 |
1.8,0.7 |
2.5,0.7 |
4.5,0.7 |
6,0.7 |
Grading fineness(μm) |
1-30 |
1-30 |
1-30 |
1-30 |
1-30 |
System air volume(m³/h) |
600 |
2600 |
4000 |
7500 |
9000 |
Rotor power(kw) |
3 |
5.5 |
7.5 |
11*3 |
15*3 |